Aiming to be a Global Specialty Pharma
Tatsuya Okamoto
Corporate Officer / Executive Director, Clinical Development
Aiming to be a Global Specialty Pharma

Tatsuya Okamoto
Corporate Officer / Executive Director, Clinical Development
—Pipeline Expansion, Maximizing Product Value, and Accelerating Global Development—
We are moving forward with clinical development in the key areas of oncology, immunology & inflammation and neurology, and focusing on pipeline reinforcement (strengthening), maximizing product value, and accelerating global development. To strengthen the pipeline, we are enhancing our ability to conduct trials in order to establish proof of concept (POC) at an early stage, and also incorporating various strategies to improve the accuracy of result interpretation.
For products in the pipeline that have already been launched, we are maximizing product value by adding supplementary indications and developing new combination therapies to meet the diverse unmet needs that still exist.
Accelerating development speed—obtaining approval faster—is extremely important to maximize the value of new drug candidates. We maximized the development functions of Deciphera in the U.S. and Europe, which newly joined our Group last year, and will steadily proceed with international joint trials to obtain global approval as soon as possible.
Clinical Development
Reinforcement of pipelines
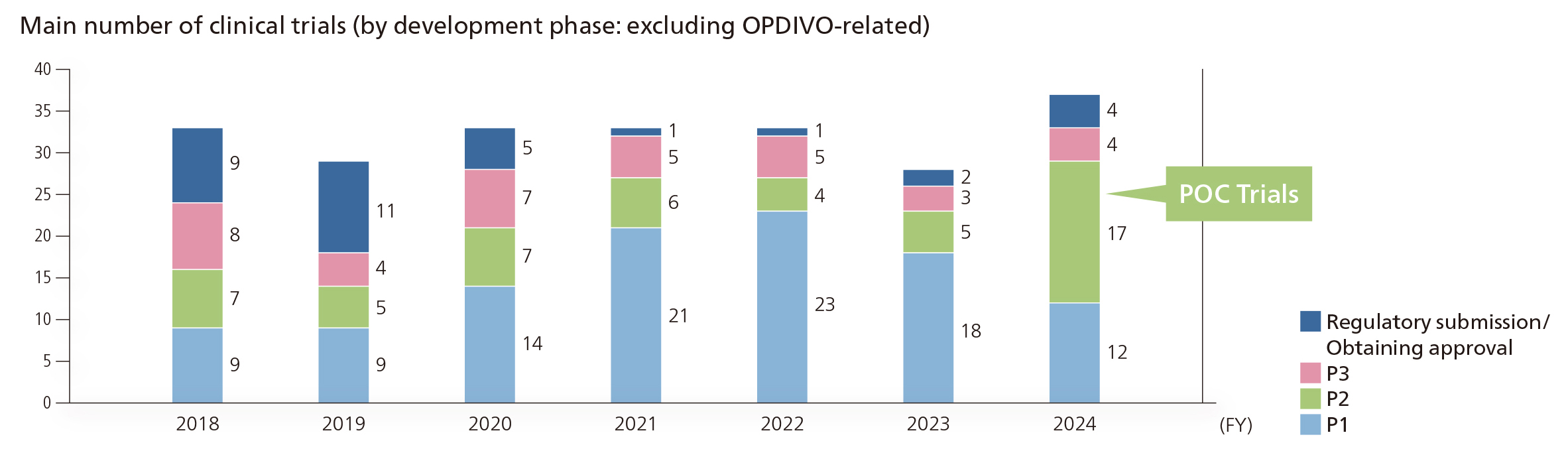
In clinical development, we are committed to achieving a qualitative enhancement of our pipeline. This refers to establishing a robust portfolio of projects with validated proof of concept (POC), and realizing a state in which numerous pivotal studies are actively underway. To deliver drug candidates created by our drug discovery research and those acquired through licensing activities as quickly as possible to patients suffering from diseases around the world, it is necessary to strengthen the quality of our pipeline. Accordingly, it is important to establish POCs early and to build and operate systems that allow for the parallel implementation of clinical trials targeting multiple diseases simultaneously, thereby shortening the time from a transition to the clinical stage, to the establishment of the POC. We are also actively working to utilize biomarkers based on Translational Research along with real-world data and clinical data obtained in-house as a way to accurately establish POC in clinical trials that are as compact as possible.
Maximization of product value
To enhance the product value of compounds, we are also working on expanding the indications of existing marketed products. In addition to expanding the indications of existing marketed products, we are also continuing work to develop combination therapies that can be used at earlier stages of treatment and that improve treatment efficacy in order to meet unmet medical needs. Furthermore, we are implementing measures to reduce the burden on patients by considering more convenient methods of administration.
Toward the Realization of the Growth Strategy
Acceleration of global clinical development
Up until now, with the exception of Korea and Taiwan, where we have established our own sales systems, we have licensed out to partner companies the clinical development and sale of drug candidates discovered in-house. However, from now on we intend to ourselves deliver the drug candidates we have discovered and developed ourselves to patients in the U.S. and Europe, the world’s largest markets. To achieve this goal, we have been working to strengthen and develop a system capable of handling all clinical development processes, including not only conducting clinical trials in the U.S. and Europe, but also everything from regulatory submission to obtaining approval. With Deciphera, which has experience with regulatory submissions and obtaining approvals in the U.S. and Europe, joining our Group last year, we will consolidate clinical development functions in Deciphera and accelerate global development even further.